The onset of the COVID-19 pandemic brought unprecedented challenges to personal and professional lives globally. Interpersonal human interaction was suddenly stripped from our everyday lives, visits with loved ones were put on hold, and what we once considered “normal life” was instantly redefined. Beyond the personal impacts came significant disruption in global supply chains, shortages in raw materials, and the inability to obtain critical PPE and other medical technologies that were the backbone to solving the pandemic’s greatest challenges.
Innovation was required to turn the obstacles presented by the pandemic into opportunities –opportunities for individuals and businesses to bring their unique skillsets and resources to solve critical challenges and better serve our global community. The innovation of individuals and businesses throughout the pandemic illustrated how flexibility and adaptability can lead to breakthroughs that leave a positive impact on the world.
One such business that embodied this innovation throughout the pandemic was Nectar, a product development and industrial design company based out of Long Beach, California. Nectar used its long history of developing integrated products across multiple sectors combined with its deep bench of engineers to design and build a low-cost ventilator system to support the pandemic response, with an end goal of serving many underprivileged regions around the world.
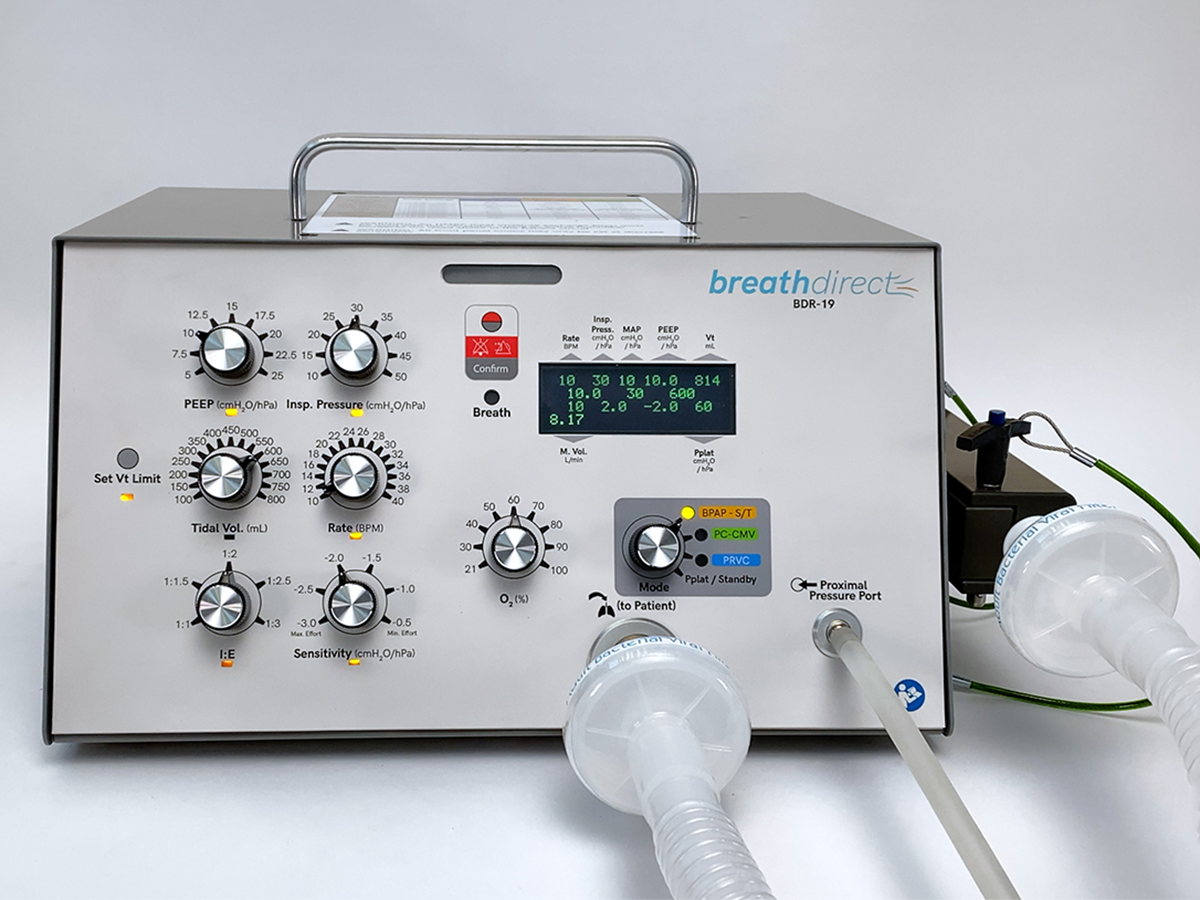
This work resulted in the launch of a new company, Breath Direct, which focuses on building and launching ventilator devices for critical care facilities across the globe. After months of tireless work, Breath Direct developed the BDR-19, a cost-effective and highly capable ventilator that meets the daily needs of resource-constrained critical care facilities who desire a unit that bridges the gap between high-acuity, moderate acuity, and makeshift care settings.
A critical goal for the BDR-19 ventilator project was to produce a high performing, high quality device that came at a cost-competitive standpoint so that Breath Direct could deploy the device to government agencies and hospitals, including those in disadvantaged areas globally. This meant the device needed to be affordable, easily deployable, quick to set up, and require minimal training.
To achieve this cost-efficient yet high-quality ventilator, Breath Direct partnered with Whitman Controls, a manufacturer of custom-engineered pressure, vacuum, level, and temperature sensor solutions based in Bristol, Connecticut. Whitman Controls leveraged its over 50 years of experience in the industry to co-develop with Breath Direct a customized pressure switch that would meet the exact performance and electrical specifications desired by the BDR-19, while still providing the purity required in any medical technology. A custom variation of Whitman’s W117G Ultra-Pure Stainless Steel Pressure Switch provided high caliber and high accuracy performance at a price that allows the BDR-19 to be the ventilator technology of choice in its target markets. It was the ultimate solution.
“The partnership we built with CEO Darren Saravis and the Breath Direct team epitomizes the relationships we look to establish at Whitman Controls” said current President, William Brame, “our team of mechanical and electrical engineers worked intimately with their team to design and build a highly-effective custom pressure switch solution, and importantly one that comes at a price point that allows Breath Direct to cost-effectively serve the critical needs of their end customers.”
Breath Direct’s BDR-19 is being marketed under an FDA Emergency Use Authorization (EUA) and is currently awaiting FDA 510k approval to market the device both domestically and internationally, but that has not restricted the team from having targeted discussions with partners in India, Africa, and other disadvantaged countries who are in dire need of this type of cost-effective critical equipment. Despite the numerous headwinds presented by the COVID-19 pandemic, Nectar Product Development and Breath Direct were able to leverage their expertise, and those of partners such as Whitman Controls, to achieve their goal of putting critical medical device technologies in the hands of the global communities that need it most.